Technologies
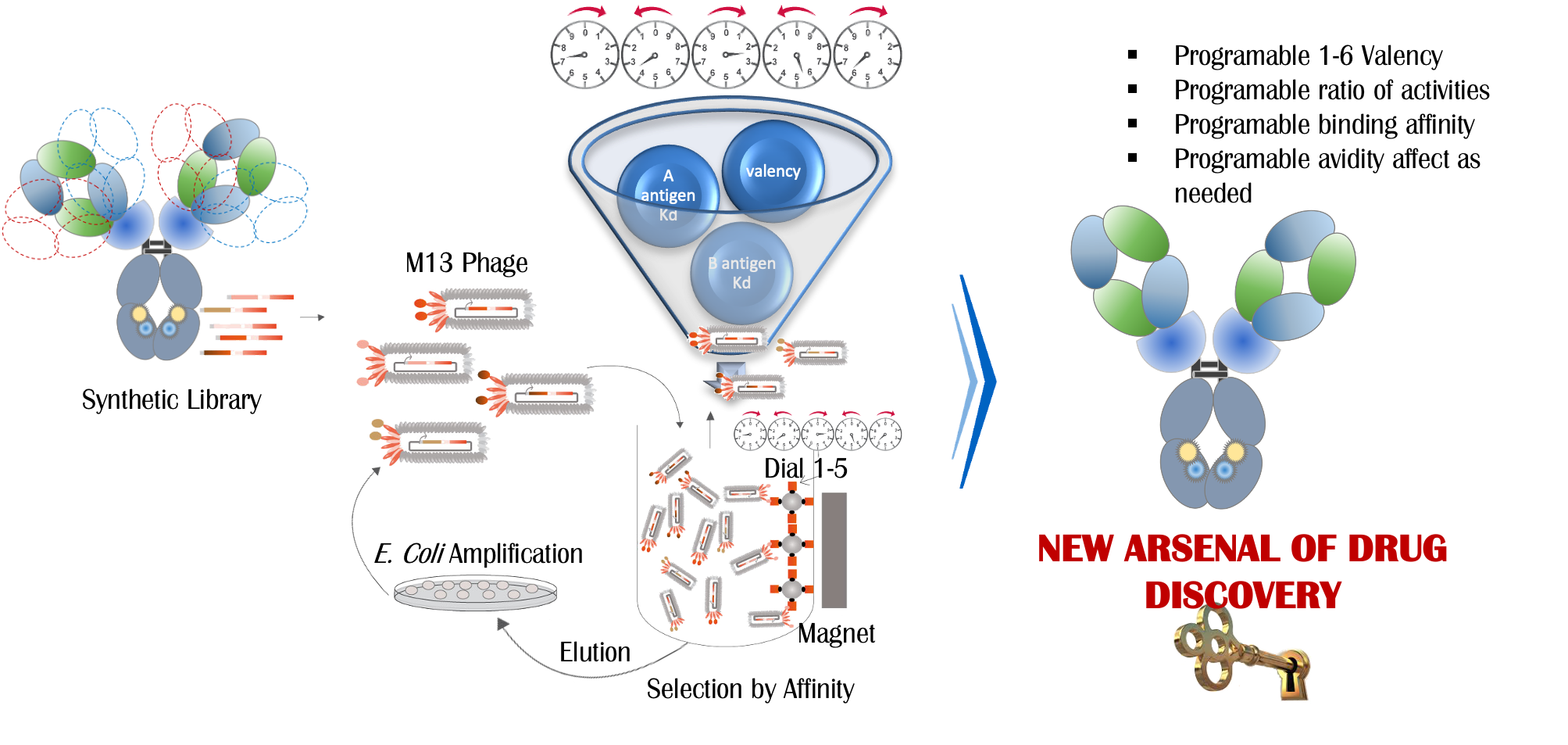
SAILINGTM
From target validation to cGMP manufacturing – an integrated approach to a path of least resistance
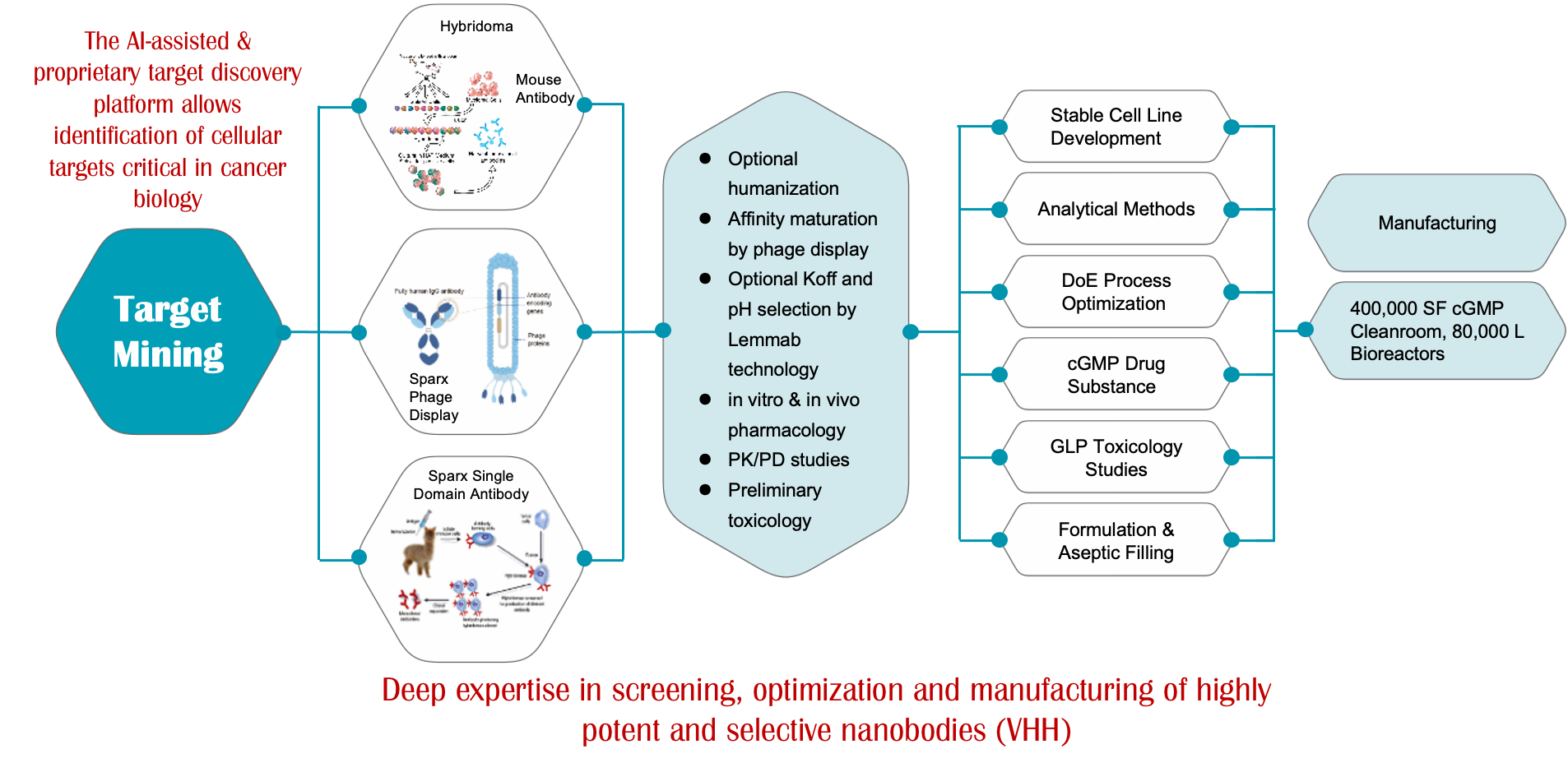
SynMabTM
Design, Selection, Optimization, CLD, CMC, and IND Filing, All within 18 Months
Deep expertise in screening, optimization and manufacturing of highly potent and selective nanobodies (VHH)
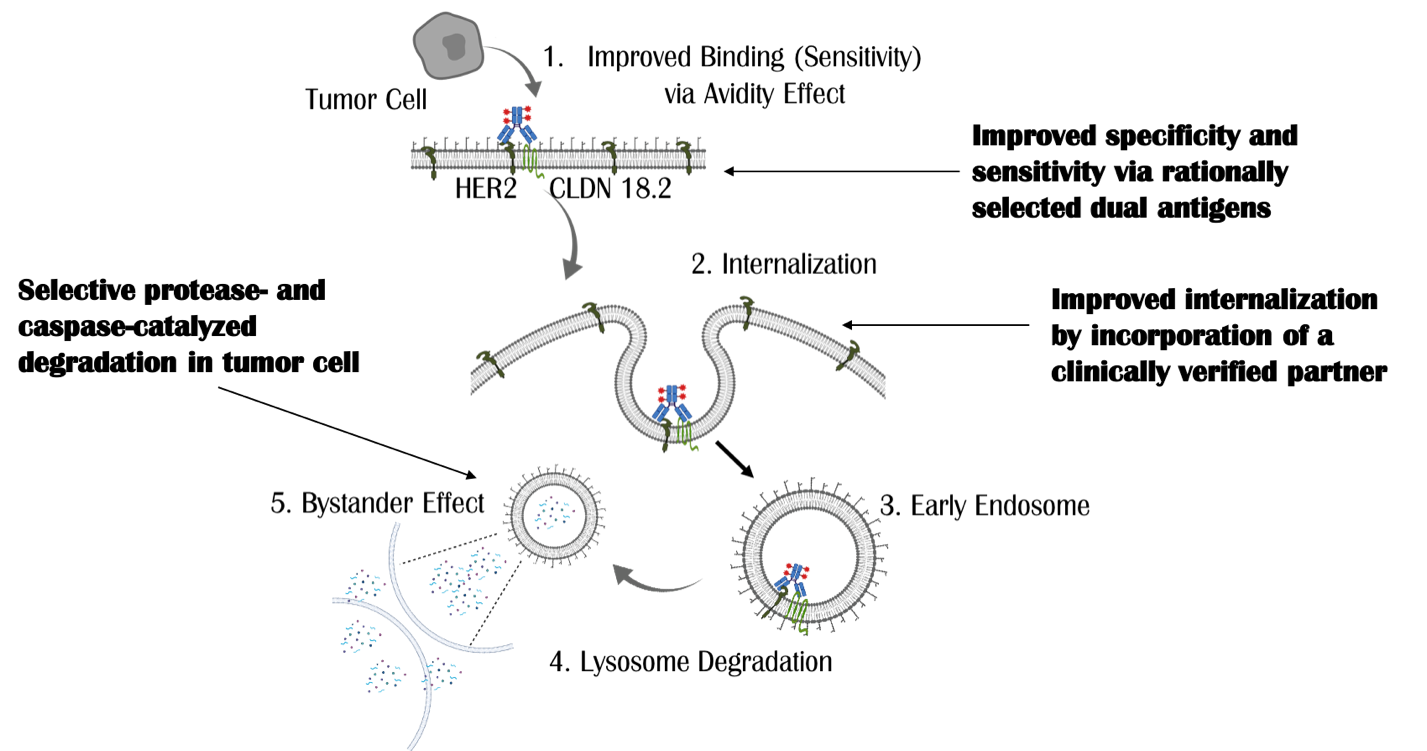
Universally empowering ADCs with high specificity, sensitivity and pharmacological activity
Get in Touch with SparX
- 700 E. Business Center Dr. Mount Prospect, IL 60056
- +1 847 257 7910 (Direct)
- +1 224 580 2979 (extension)
- info@sparxbio.com